|
4/5/2024 0 Comments Density of water tableTemperature O CPressure bar c p kJ/kgK kg/m3 10 6 Pas 10 6 m2/s 0.0 1. This is why seawater at Antarctica is very salty. Table of water properties - temperature, pressure, density, viscosity. The salt that is rejected forms brine beneath the ice and becomes more and more salty until it becomes so dense that it sinks, displaces less dense seawater that moves to the surface. As ice forms in the sea, the salt cannot form part of the ice crystal so the ice is almost pure water.The salinity of seawater is about 3.5% and it freezes at about -1.9☌.Both below and above this temperature, the density of water is lower. The addition of salt to water makes a solution that is denser than fresh water – it freezes at a lower temperature. The density of water reaches its maximum around 4☌. 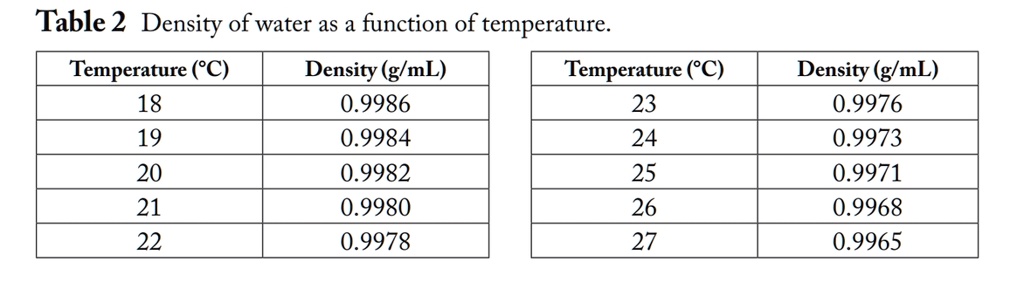
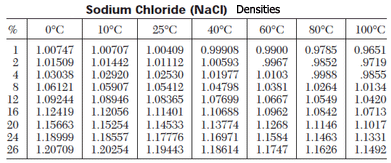
The hydrogen of the water molecule is attracted to chlorine ions and the oxygen to the sodium ions. Density of Water in a few select units of density measurement: Density of Water g cm3 1 g/cm³ Density of Water g ml 1 g/ml Density of Water g mm3 0. This allows the sodium and chlorine ions to be pulled apart by the water molecules. Table 14.2 shows the density of water in various phases and temperature. A standard table lists the values for the density of liquid water. The density of solids and liquids normally increase with decreasing temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
